Chemical Bonding
- Miguel Aveiro
- Jun 13, 2020
- 5 min read
Updated: Feb 28

In this article, we'll learn about:
2.
Metals and non metals
Covalent bonds
Ionic bonds
How metal atoms are bound together
Lattice structures
2
Metals and non metals
Elements can be either metals or non metals. Undoubtedly, you would have heard of some metals: iron, copper, nickel, gold, silver, platinum. They are found in different quantities inside the Earth (gold, silver and platinum are rarer than most of the others and are considered very valuable). I'll do another article on materials, including metals, so I won't go into more detail about them here.
Let's take another look at the periodic table:

Basically, all the elements are metals except for the ones in green, yellow or light blue, which are non metals and the ones in dark green are metalloids. Don't worry about metalloids for now.
For the benefit of those who have difficulty seeing colours, the non-metals are hydrogen (H) on the top left and then on the right you have: carbon (C), nitrogen (N), oxygen (O), phosphorus (P), sulphur (S) and selenium (Se); and groups 17 and 18, except for tenessine (Ts) and oganesson (Og). The metalloids are: boron (B), silicon (Si), geranium (Ge), arsenic (As), antimony (Sb) and tellurium (Te).
How atoms bind together depends on whether they are metals or non metals.
Covalent bonds
Covalent bonds are the chemical bonds between non-metals and non-metals. They are also known as molecular bonds because the atoms form molecules via these bonds. These are the bonds we've seen already: they involve a free electron from one atom pairing up with a free electron from another atom, so that the two atoms share their electrons.
The simplest example of a covalent bond is between two hydrogen atoms:

Here is a water molecule (H2O) which has an oxygen atom sharing electrons with two hydrogen atoms:

Covalent bonds are the strongest type of bonds, because the shared electrons are very difficult to separate.
Ionic bonds
Ionic bonds are the chemical bonds between metals and non metals. They are strong bonds but not as strong as covalent bonds. Metals and non metals don't share electrons with each other in the same way. Instead, what happens is a free electron from a metal atom is attracted to the non metal atom, so that the electron pairs up with a free electron in the non metal atom.
We'll see this in the simplest example, with lithium (Li) and hydrogen (H):

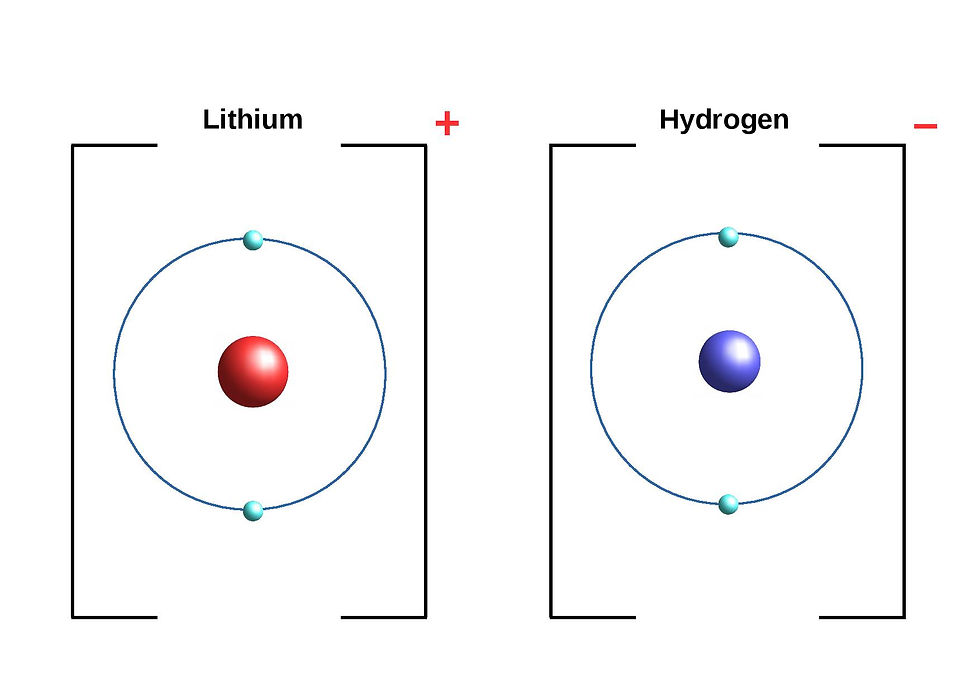
So we end up with lithium having one less electron and hydrogen with one more electron.
Before binding, lithium has three protons which are positive and three electrons which are negative. This means that the atom, as a whole, doesn't have a charge: the positive and negative charges cancel each other out, so it's neutral. But when it loses an electron as it bonds with hydrogen, it has two electrons and three protons. Now the atom, as a whole, is positively charged.
Hydrogen, before binding, has one electron and one proton, so again, like lithium, it's neutral. After binding, the hydrogen atom, as a whole, ends up negative as there are now two electrons and one proton.
We call these charged atoms ions. The positive ions are known as cations and the negative ions are anions. We represent these with brackets around the ions and + and - signs indicating which one is positive and which one is negative.
What's actually happening, is the hydrogen atom is attracting the electron from the lithium atom. Electronegativity is a measure of both an element's tendency to do this and how much it does (or in other words, how strongly it attracts the electron or electrons). So, as hydrogen is more electronegative, it pulls the electron towards it. The electron is shared, like in a covalent bond, but it's closer to the hydrogen atom than the lithium atom. With an ionic bond, the metal always ends up as a cation (positive) and the non-metal will be an anion (negative).
Some scientists, such as biologists, may call the combination of a metal and non metal a molecule. However, chemists use the term molecule only for covalently bound non metal atoms with other non metal atoms. A metal atom with a non metal atom is an ionic compound.
The term compound refers to two elements bound together. A non metal element with another non metal element (or you could say atom instead of element) makes a covalent compound.
What about a metal atom with another metal atom?
A metal atom does not form a covalent or ionic bond with another metal atom. The atoms are still bound together, however, and metals are usually very strong and tough.
What happens is the metal forms cations with the electrons moving around freely between them. So the cations bind with these electrons and form a network called a lattice:

Other lattice structures
It's not only metal atoms binding with other metal atoms that form lattice structures, but metals with non metals and non metals with non metals.
Carbon is an element that can form different types of lattices. These different types are referred to as allotropes. These lattices involve the atoms held together via covalent bonds.
Graphite is the form of carbon that is used to make pencils. The carbon atoms form flat layers with spaces in between. Each individual layer is called graphene.

Each carbon atom forms a bond with three others. Most of them form a bond with an atom of another layer and all the layers link up. The bonds between graphene layers, however, are not as strong as within the layers themselves. Therefore, the layers can break away and so graphene from the pencil ends up on the surface of paper.
Carbon can also form diamond, which consists of atoms held together in such a way as to make a very strong material (in fact, it's the strongest known natural substance in the universe). Have a look:

It's not the individual atom to atom bonds that are so strong, as that would make diamond just as strong as graphite, but it's the way they are arranged. You could imagine cutting graphite with a knife that's only a layer of molecules in thickness. The blade can slice between the layers. But even a knife as sharp as that would have a hard time against diamond which doesn't have such layers. The pattern of bonds forms an incredibly tough and durable structure.
So there you have a little more information about chemical bonds. There is more that I could go into, but as this is a Key Article, the knowledge contained here is background for all the areas of science, not just chemistry. Other types of bonds and interactions between atoms and molecules are topics which are important for chemistry but not so much other areas of science, unless you get into Level 4 or maybe Level 3 areas.

Comments